A brief but valuable blog post:
http://www.scientificamerican.com/blog/post.cfm?id=the-antidepressant-reboxetine-a-hea-2010-11-30&sc=emailfriend
--from SciCurious at the Scientific American blog offers some very valuable insights on the recent failure of the antidepressant reboxetine, as we previously reviewed:
http://brodyhooked.blogspot.com/2010/11/pfizers-reboxetine-latest-in-series-of.html
What SciCurious adds to our understanding is that the failure of reboxetine is an even bigger setback for research and drug development than would be evident from the failure of a single drug. The problem is that based on everything you can do in the test tube and in lab rats, reboxetine should be the best antidepressant since sliced bread. It jumps thru all the hoops that scientists have long relied on to screen substances for possible antidepressant effects in humans. The fact that a drug passes all these tests and then doesn't work in humans means that the tests are now seriously called into question--as are the basic scientific theories of neurotransmitter function that the tests are based on. In a previous post I briefly alluded to Leo and Lacasse's excellent work in exposing the flaws in the serotonin theory of depression:
http://brodyhooked.blogspot.com/2009/03/jama-editors-need-to-come-down-off.html
It now seems even more likely that we need to go back to the drawing board in figuring out what exact sort of "chemical imbalance," if any, depression really represents, and how that relates to finding better drugs for it. This example simply highlights what really hard work it is to find good new drugs. It's sad that an industry that used to be really good at finding these drugs seems so seriously to have gotten out of the business--as reviewed in the most recent post befoere this.
Friday, January 28, 2011
Sunday, January 23, 2011
Drug Discovery: Some Imcompetent Reflections
The Feds have now made it official: The pharmaceutical industry doesn't know how to discover useful new drugs.
As reported by Gardiner Harris in the New York Times:
http://www.nytimes.com/2011/01/23/health/policy/23drug.html
--the government has run out of patience with the slow pace of drug discovery and plans to invest $1B in a new National Center for Advancing Translational Sciences within the NIH, specifically to do some of the drug development that private industry once did but, apparently, no longer does.
Now, I am not a research scientist, nor do I play one on TV, and I have no idea how to discover new drugs. Despite my near-total incompetence on these matters I will offer some reflections, many of which were previously offered in HOOKED or in previous blog entries.
First, what is wrong with the current state of industry drug discovery? This has a two-part answer--new drugs are quite hard to discover these days, and Pharma has essentially gotten itself out of the drug discovery business.
The big reason drugs are hard to discover is what I've previously quoted economist Uwe Reinhardt about, the paucity of low-hanging fruit. Once we have a basic understanding of what causes various common diseases, we can figure out what sorts of molecules might make useful drugs to treat them, but after a while, we have discovered most of the easy-to-find molecules that produce drugs that are both safe and effective. The as-yet-undiscovered molecules are fewer in number and much harder to find, or else more likely to cause adverse reactions while being no better than existing drugs. Until such time as we advance our basic understanding of disease, we can expect a lull in the pace of discovery. It seems as if we're more or less in one of those lulls just now. (If you think about all the things a molecule has to do inside of and in interaction with the human body to become a safe and effective drug, it's a miracle there are any useful drugs at all.)
Now, even given that good new drugs are hard to find, it remains the fact that Pharma seems ill-equipped just now to find them anyway. There are three main reasons for this so far as I can figure.
Clearly the new Center is supposed to be a part of the love affair that the NIH has declared with the idea of "translational science." Depending on who's talking, you can get very different ideas about what "translational science" is. On the positive side, translational science seems to mean having scientists in different fields form interdisciplinary teams, communicate with each other, and exchange ideas throughout the course of research. If you try to implement this model, you find that it's hard to do because the average scientist was simply not trained in this way. That in turn suggests the need to train future scientists more in the translational setting from the get-go. All that, I suggest, is for the good, though unlikely to produce quick results.
On the negative side, sometimes it sounds as if what "translational science" means (in terms of moving research findings more quickly "from the bench to the bedside") is "let's do what they do in private industry" or "let's do what will make us the most money soonest." If the industry R&D model is a bust, then why the heck model our science after it?
To my poor ignorant mind, it seems that ultimately, we'll discover new, useful drugs to the extent that we invest in more basic science research and learn more about the mechanisms of disease. That's not sexy, and it certainly cannot be preprogrammed to suit Wall Street's quarterly earning reports. But it seems to explain, for instance, why we seem poised for real advances in cancer drugs in the next decade or so, because we now know a lot more about tumor cell genetics and enzymology. Does this mean that we won't occasionally get lucky and discover good drugs purely by serendipity? Of course that can happen; what about aspirin and penicillin? But we probably can't count on that as our best and sole approach.
As reported by Gardiner Harris in the New York Times:
http://www.nytimes.com/2011/01/23/health/policy/23drug.html
--the government has run out of patience with the slow pace of drug discovery and plans to invest $1B in a new National Center for Advancing Translational Sciences within the NIH, specifically to do some of the drug development that private industry once did but, apparently, no longer does.
Now, I am not a research scientist, nor do I play one on TV, and I have no idea how to discover new drugs. Despite my near-total incompetence on these matters I will offer some reflections, many of which were previously offered in HOOKED or in previous blog entries.
First, what is wrong with the current state of industry drug discovery? This has a two-part answer--new drugs are quite hard to discover these days, and Pharma has essentially gotten itself out of the drug discovery business.
The big reason drugs are hard to discover is what I've previously quoted economist Uwe Reinhardt about, the paucity of low-hanging fruit. Once we have a basic understanding of what causes various common diseases, we can figure out what sorts of molecules might make useful drugs to treat them, but after a while, we have discovered most of the easy-to-find molecules that produce drugs that are both safe and effective. The as-yet-undiscovered molecules are fewer in number and much harder to find, or else more likely to cause adverse reactions while being no better than existing drugs. Until such time as we advance our basic understanding of disease, we can expect a lull in the pace of discovery. It seems as if we're more or less in one of those lulls just now. (If you think about all the things a molecule has to do inside of and in interaction with the human body to become a safe and effective drug, it's a miracle there are any useful drugs at all.)
Now, even given that good new drugs are hard to find, it remains the fact that Pharma seems ill-equipped just now to find them anyway. There are three main reasons for this so far as I can figure.
- The drug industry has been into me-too drugs in a big way. Rather than go after a whole new class of drugs, it's much simpler and safer to tweak an existing molecule slightly and then try to market the result as the "new purple pill" or whatever. (The wimps at the Patent Office enable this by granting patents in apparent disregard for the criterion of non-obviousness, which is one of the standard legal requirements for patents. I am reasonably sure that if I could afford to hire the right attorneys, I could take out a patent on the period at the end of this sentence.)
- The industry has outsourced all risk to biotech start-ups. These biotech companies are often based on new and even wacky ideas. At least 9 out of 10 will go bust. The big drug companies can wait around and then buy out the occasional, rare biotech company whose idea actually pans out and looks like it could turn into a drug.
- The third reason is actually at the root of the first two as well. The companies have put their bean counters almost totally in charge of the research agenda. If a line of research seems like it's not going to turn into a billion bucks in sales by next week, they axe it--even if, pursued for a couple more years, it could have produced sterling results.
Clearly the new Center is supposed to be a part of the love affair that the NIH has declared with the idea of "translational science." Depending on who's talking, you can get very different ideas about what "translational science" is. On the positive side, translational science seems to mean having scientists in different fields form interdisciplinary teams, communicate with each other, and exchange ideas throughout the course of research. If you try to implement this model, you find that it's hard to do because the average scientist was simply not trained in this way. That in turn suggests the need to train future scientists more in the translational setting from the get-go. All that, I suggest, is for the good, though unlikely to produce quick results.
On the negative side, sometimes it sounds as if what "translational science" means (in terms of moving research findings more quickly "from the bench to the bedside") is "let's do what they do in private industry" or "let's do what will make us the most money soonest." If the industry R&D model is a bust, then why the heck model our science after it?
To my poor ignorant mind, it seems that ultimately, we'll discover new, useful drugs to the extent that we invest in more basic science research and learn more about the mechanisms of disease. That's not sexy, and it certainly cannot be preprogrammed to suit Wall Street's quarterly earning reports. But it seems to explain, for instance, why we seem poised for real advances in cancer drugs in the next decade or so, because we now know a lot more about tumor cell genetics and enzymology. Does this mean that we won't occasionally get lucky and discover good drugs purely by serendipity? Of course that can happen; what about aspirin and penicillin? But we probably can't count on that as our best and sole approach.
Monday, January 17, 2011
Composite Endpoints: Another Way to Fool Journal Readers
In our ongoing series, "How to Make Silk Purses out of Sows' Ears when Conducting and Reporting Research," we now cite a recent paper by authors from the Nordic Cochrane Center in Copenhagen and the Dartmouth policy center (subscription required).
When you do a randomized controlled trial, you are supposed to say up front what your endpoint is going to be. For example, if you're studying a new cholesterol-lowering drug, then good endpoints to study would be heart attack, stroke, and death. (Not-so-good endpoints to study would be level of cholesterol. This is the infamous "surrogate endpoints" where we simply assume that if you reduce cholesterol, then of course you must be reducing the number of heart attacks, strokes and deaths later on. The problem with surrogate endpoints is that they've shown many times for many different diseases that these assumptions may be bogus. Drug companies love surrogate endpoints because the studies are cheap; you can show that your drug lowers cholesterol in a couple of months, where it would take years to show that it reduces really important endpoints, if it even does. Plus they have armies of reps to convince docs that the surrogate endpoints really mean something.)
Sometimes, for various statistical and methodological reasons, the investigators may not report heart attack, stroke, and death separately, or not only those. They may also calculate a combined score that reflects whether an individual subject experienced any of the three outcomes. This is called a composite endpoint. A composite endpoint that reflects the risk of stroke, heart attack, and/or death with and without the drug is probably a reasonably valid endpoint, since all of these are bad and you could argue they are all really bad. Ideally, the investigators would report the three endpoints separately as well as the composite. That way if any armchair bean-counters thought they were up to somthing, they could easily double check.
Enter the present set of authors, headed by Gloria Cordoba. They looked at 40 clinical trials published in 2008 that reported composite endpoints. Nearly 3/4 of them were about cardiovascular stuff (like my example above) and 83% had either total or partial industry funding.
To cut to the chase, Cordoba and colleagues found a lot of funny business, such as:
Final conclusion: beware composite endpoints. If they are not actually trying to make a silk purse out of the ear of this pig, they are probably at the very least putting lipstick on it.
Cordoba G, Schwartz L, Wolosin S, et al. Definition, reporting, and interpretation of composite outcomes in clinical trials: a systematic review. BMJ 341:c3940, Aug. 18, 2010.
When you do a randomized controlled trial, you are supposed to say up front what your endpoint is going to be. For example, if you're studying a new cholesterol-lowering drug, then good endpoints to study would be heart attack, stroke, and death. (Not-so-good endpoints to study would be level of cholesterol. This is the infamous "surrogate endpoints" where we simply assume that if you reduce cholesterol, then of course you must be reducing the number of heart attacks, strokes and deaths later on. The problem with surrogate endpoints is that they've shown many times for many different diseases that these assumptions may be bogus. Drug companies love surrogate endpoints because the studies are cheap; you can show that your drug lowers cholesterol in a couple of months, where it would take years to show that it reduces really important endpoints, if it even does. Plus they have armies of reps to convince docs that the surrogate endpoints really mean something.)
Sometimes, for various statistical and methodological reasons, the investigators may not report heart attack, stroke, and death separately, or not only those. They may also calculate a combined score that reflects whether an individual subject experienced any of the three outcomes. This is called a composite endpoint. A composite endpoint that reflects the risk of stroke, heart attack, and/or death with and without the drug is probably a reasonably valid endpoint, since all of these are bad and you could argue they are all really bad. Ideally, the investigators would report the three endpoints separately as well as the composite. That way if any armchair bean-counters thought they were up to somthing, they could easily double check.
Enter the present set of authors, headed by Gloria Cordoba. They looked at 40 clinical trials published in 2008 that reported composite endpoints. Nearly 3/4 of them were about cardiovascular stuff (like my example above) and 83% had either total or partial industry funding.
To cut to the chase, Cordoba and colleagues found a lot of funny business, such as:
- In at least 4/5 of the studies, the composite was made up of individual endpoints that the authors judged not to be equally important. My pal Jerry Hoffman of Primary Care Medical Abstracts fame (and thanks to PCMA for calling my attention to this study) uses as his standard satirical example "stroke, heart attack, and hangnail." The point here is that the company's drug may not do any better at reducing stroke or heart attack, but it may be statistically superior at reducing hangnail. Instead of saying, "Our drug reduced hangnails," the authors get to announce, "Our drug substantially reduced the composite endpoint that included stroke and heart attack (and oh yes, by the way, hangnails were somewhere in there too)." They hope you won't notice that the hangnails accounted for all the real drug benefit and the stroke and heart attack not at all.
- In 10% of the trials, the authors flat out admitted that they'd made up to composite endpoint only after all the data were in. That's the equivalent of shooting your arrow at a wall, and then drawing the bull's-eye around the arrow wherever it happened to hit.
- In 1/3 of the trials, the definition of the composite endpoint shifted from the abstract to the methods to the results sections of the paper. This is a strong hint that the painting-the-bull's-eye trick was going on though these authors were not honest enough to say so.
- In as many as 82% of the articles, the reader was not reminded at the end that what had gotten better was the composite endpoint but not necessarily the individual components--in other words, major spin.
Final conclusion: beware composite endpoints. If they are not actually trying to make a silk purse out of the ear of this pig, they are probably at the very least putting lipstick on it.
Cordoba G, Schwartz L, Wolosin S, et al. Definition, reporting, and interpretation of composite outcomes in clinical trials: a systematic review. BMJ 341:c3940, Aug. 18, 2010.
Saturday, January 15, 2011
Sunshine and Advocacy Groups
Another recent paper in American Journal of Public Health sheds some useful light on the problem of health advocacy groups getting funding from Pharma (subscription required). The Rothmans and their Center on Medicine as a Profession at Columbia have written a number of sterling papers, so I am sorry to have to add a couple of reservations to my review of their new paper.
At issue here are the patient advocacy organizations, which we docs when grousing among ourselves sometimes call "disease of the month clubs" while acknowledging all the great work they do in providing support and services to patients and their families who feel overwhelmed with diseases like cancer, muscular dystrophy, and diabetes. The not-so-great work they do is lobbying, which is very good in keeping pressure on the government to fund medical research, but which also risks distorting that research by pushing money into politically popular causes rather than what's most needed or what areas of science are really primed for rapid advance.
In the past both drug companies and advocacy groups have been pretty tight-lipped about how much and what sort of funding Pharma provides for these groups. In HOOKED I described some advocacy groups which were so solely the creatures of the drug firms that they counted as "astroturf" (fake grass roots). More commonly these groups have legitimate grass roots support and really do represent patients and families; but the drug companies for that very reason see them as prime targets for marketing influence. A thousand real patients picketing the state capitol is worth more than a gaggle of drug company lobbyists.
After Congress made a lot of noise about passing a sunshine law (which has now become official until such time as the Republicans succeed in repealing the health reform law), Eli Lilly became the first drug company voluntarily to create a public web database of all their gifts and grants. The Rothmans therefore used this database to compare what Lilly said they gave, to what the various adocvacy groups admitted that they received.
Their results: First, they noticed that Lilly was highly selective in giving grants. If Lilly did not have a drug for your disease, sorry, your organization did not get any grants from them. Comment: Now, I suppose for thoroughness they had to do this step of the study, but really--isn't this a "well, duh"? What part of "marketing" don't we understand?
The next revelation is the real meat of the paper, showing that the websites for these organizations only acknowledged receiving Lilly money a minority of the time; and virtually never say just how much money it was.
Now, this is an important finding, but we also sadly need to qualify it some. I think this study represents a single moment in time, and the times they are a-changin'. The old way of doing business was to keep all this under your hat. Then with Lilly's website, a new trend started, of transparency (under the gun of the Feds no doubt). You'd naturally expect that as more and more companies started to come clean on their payouts, then the recipients of the largesse will start to realize that they look pretty silly if they try to keep this under wraps, and will gradually change their own disclosure practices. And indeed, in recent years groups like the mental health people (NAMI) and the American Diabetes Assn have engaged in some soul-searching about accepting so much industry money--though they have a lot of soul-searching left to do in my humble opinion. So this study will be of little use unless a follow-up is done in a few years to see what the overall trend has been.
I dunno if anyone at Columbia condescends to read this blog, but if they do, I welcome a comment in further defense of the study.
Rothman SM, Raveis VH, Friedman A, Rothman DJ. Health advocacy organizations and the pharmaceutical industry: an analysis of disclosure practices. American Journal of Public Health doi:10.2105/AJPH.2010.300027, epub Jan. 13, 2011.
At issue here are the patient advocacy organizations, which we docs when grousing among ourselves sometimes call "disease of the month clubs" while acknowledging all the great work they do in providing support and services to patients and their families who feel overwhelmed with diseases like cancer, muscular dystrophy, and diabetes. The not-so-great work they do is lobbying, which is very good in keeping pressure on the government to fund medical research, but which also risks distorting that research by pushing money into politically popular causes rather than what's most needed or what areas of science are really primed for rapid advance.
In the past both drug companies and advocacy groups have been pretty tight-lipped about how much and what sort of funding Pharma provides for these groups. In HOOKED I described some advocacy groups which were so solely the creatures of the drug firms that they counted as "astroturf" (fake grass roots). More commonly these groups have legitimate grass roots support and really do represent patients and families; but the drug companies for that very reason see them as prime targets for marketing influence. A thousand real patients picketing the state capitol is worth more than a gaggle of drug company lobbyists.
After Congress made a lot of noise about passing a sunshine law (which has now become official until such time as the Republicans succeed in repealing the health reform law), Eli Lilly became the first drug company voluntarily to create a public web database of all their gifts and grants. The Rothmans therefore used this database to compare what Lilly said they gave, to what the various adocvacy groups admitted that they received.
Their results: First, they noticed that Lilly was highly selective in giving grants. If Lilly did not have a drug for your disease, sorry, your organization did not get any grants from them. Comment: Now, I suppose for thoroughness they had to do this step of the study, but really--isn't this a "well, duh"? What part of "marketing" don't we understand?
The next revelation is the real meat of the paper, showing that the websites for these organizations only acknowledged receiving Lilly money a minority of the time; and virtually never say just how much money it was.
Now, this is an important finding, but we also sadly need to qualify it some. I think this study represents a single moment in time, and the times they are a-changin'. The old way of doing business was to keep all this under your hat. Then with Lilly's website, a new trend started, of transparency (under the gun of the Feds no doubt). You'd naturally expect that as more and more companies started to come clean on their payouts, then the recipients of the largesse will start to realize that they look pretty silly if they try to keep this under wraps, and will gradually change their own disclosure practices. And indeed, in recent years groups like the mental health people (NAMI) and the American Diabetes Assn have engaged in some soul-searching about accepting so much industry money--though they have a lot of soul-searching left to do in my humble opinion. So this study will be of little use unless a follow-up is done in a few years to see what the overall trend has been.
I dunno if anyone at Columbia condescends to read this blog, but if they do, I welcome a comment in further defense of the study.
Rothman SM, Raveis VH, Friedman A, Rothman DJ. Health advocacy organizations and the pharmaceutical industry: an analysis of disclosure practices. American Journal of Public Health doi:10.2105/AJPH.2010.300027, epub Jan. 13, 2011.
Diminishing Returns: Improving Health vs. Selling Drugs
In the previous post I laid out the Inverse Benefit Law that Donald Light and I recently proposed. Another insightful article recently appeared that demonstrates further how the basic mechanisms work that cause a clash between drug marketing and public health:
http://www.jabfm.org/cgi/content/full/23/3/371
Dr. James W. Mold and colleagues from U. Oklahoma Dept. of Family and Preventive Medicine address a phenomenon that is becoming common as new guidelines are promulgated with management strategies for chronic diseases (often, today, written by authors or organizations with significant financial conflicts of interest with Pharma). If docs follow the guidelines rigorously, they will feel duty bound to attempt numerous risk reduction strategies for each patient. The authors construct a hypothetical illustrative case of a 65-year-old African American man diagnosed a year ago with Type 2 diabetes, whose blood pressure, hemoglobin A1C (a measure of diabetes control), and "bad" cholesterol are all higher than normal. According to standard guidelines, this patient needs at least 6 major medical or lifestyle interventions right now to reduce his risks of the major complications of diabetes such as heart attack, stroke, amputations, and kidney failure. (Given the current fetish for preventive medicine, many would be outraged at the idea that we'd skip even one of the 6.) If you read the guidelines, more often than not, these 6 interventions would all be listed as equally high priority.
Mold and colleagues tell us that the real situation is quite different and demands a much more nuanced approach. There are two reasons for this, one fairly obvious, the other less so.
The first reason is that if you look individually at the posible interventions, the 6 are not equal. They have very different effects on the reduction of risk. For instance, in our diabetic patient, taking a lo-dose aspirin daily offers twice the value (in terms of lowering the risk of a heart attack) as does lowering the "bad" cholesterol from 140 to 100.
The second reason is a matter of math. When you do intervention #1, you lower the patient's risk by a certain percentage. Let's for argument's sake pretend that all 6 interventions lower risks by the same percentage, which as we have just seen, they don't. So aspirin, let's say, reduces your risk of dying by 20%. So you add aspirin. Intervention #2 also reduces your risk by 20%, so we add that intervention. But your risk is no longer 100%; it's now down to 80% because of Intervention #1. So to calculate the new risk reduction, you need to take 20% of 80% which is only a 16% additional risk reduction. And so on each time you add a further intervention--hence the "law of diminishing returns" for risk reduction strategies.
An aside here-- what we are basically saying is that risk reduction is not additive. If you had 5 interventions, and each alone reduced your risk of dying by 20%, then if they were additive, and you did all 5, you'd become immortal. But we know the world doesn't work like that.
So back to pharmaceutical marketing. Dr. Mold and company basically are recommending that the smart primary care physician will go for the biggest bang for the buck as it were. For that hypothetical patient, if you focused on the top 3 interventions only, you'd get more than 80% of the real value of doing all 6. And if you focused on 3 instead of 6, it's much more likely that the patient would actually do them; you'd likely save a whole lot of money for both the patient and the payer; and you'd avoid the risk of medication side effects if the intervention involved a medicine. From both the individual patient-doc perspective, and the larger public health perspective, it's a smart move.
The drug industry, on the other hand, is much more likely to want docs to think that if the guidelines list 6 interventions, well doggone it, you had better do all 6. Recall that in our "diminishing turns" list for our patient with diabetes, the most effective intervention was the daily lo-dose aspirin, and the least was cholesterol lowering. To do the daily aspirin involves a cheap generic drug, that generates no profits to speak of for Pfizer or Merck or any of the big boys. (You don't see consumer ads on TV saying, "Ask your doctor if a daily generic aspirin tablet is right for you.") To get the man's cholsterol from 140 to 100 will probably take one if not two expensive statin drugs, many of which are still brand-name. Do you think the statin makers want us to think that cholesterol-lowering might be an expendible risk reduction in a smart patient-oriented strategy? Their marketing message to the doc is almost for sure to be that lowering cholesterol is every bit as important, if not more so, than any other risk reduction move.
A final tangent. Mold et al. give us pause not only about drug marketing; they also call into question the wisdom of most pay-for-performance strategies. In the abstract, the idea for paying the doc more for providing better quality care seems irrefutable. But if by "quality" we mean "meeting all the guideline targets" then we see how P4P can undermine smart practice. As the law of diminishing returns shows, you often do the best for your patient by getting close to, but not quite meeting the target--and adding more and more meds to be sure that you get to the target (whether cholesterol, or blood pressure, or blood sugar) might add very little extra benefit while greatly increasing the chances of adverse reactions. But the doc is heavily incentivized to try to meet the payment goal and not to think about what really most helps the patient.
Tip of the hat to Primary Care Medical Abstracts for including the Mold paper in their reviews.
http://www.jabfm.org/cgi/content/full/23/3/371
Dr. James W. Mold and colleagues from U. Oklahoma Dept. of Family and Preventive Medicine address a phenomenon that is becoming common as new guidelines are promulgated with management strategies for chronic diseases (often, today, written by authors or organizations with significant financial conflicts of interest with Pharma). If docs follow the guidelines rigorously, they will feel duty bound to attempt numerous risk reduction strategies for each patient. The authors construct a hypothetical illustrative case of a 65-year-old African American man diagnosed a year ago with Type 2 diabetes, whose blood pressure, hemoglobin A1C (a measure of diabetes control), and "bad" cholesterol are all higher than normal. According to standard guidelines, this patient needs at least 6 major medical or lifestyle interventions right now to reduce his risks of the major complications of diabetes such as heart attack, stroke, amputations, and kidney failure. (Given the current fetish for preventive medicine, many would be outraged at the idea that we'd skip even one of the 6.) If you read the guidelines, more often than not, these 6 interventions would all be listed as equally high priority.
Mold and colleagues tell us that the real situation is quite different and demands a much more nuanced approach. There are two reasons for this, one fairly obvious, the other less so.
The first reason is that if you look individually at the posible interventions, the 6 are not equal. They have very different effects on the reduction of risk. For instance, in our diabetic patient, taking a lo-dose aspirin daily offers twice the value (in terms of lowering the risk of a heart attack) as does lowering the "bad" cholesterol from 140 to 100.
The second reason is a matter of math. When you do intervention #1, you lower the patient's risk by a certain percentage. Let's for argument's sake pretend that all 6 interventions lower risks by the same percentage, which as we have just seen, they don't. So aspirin, let's say, reduces your risk of dying by 20%. So you add aspirin. Intervention #2 also reduces your risk by 20%, so we add that intervention. But your risk is no longer 100%; it's now down to 80% because of Intervention #1. So to calculate the new risk reduction, you need to take 20% of 80% which is only a 16% additional risk reduction. And so on each time you add a further intervention--hence the "law of diminishing returns" for risk reduction strategies.
An aside here-- what we are basically saying is that risk reduction is not additive. If you had 5 interventions, and each alone reduced your risk of dying by 20%, then if they were additive, and you did all 5, you'd become immortal. But we know the world doesn't work like that.
So back to pharmaceutical marketing. Dr. Mold and company basically are recommending that the smart primary care physician will go for the biggest bang for the buck as it were. For that hypothetical patient, if you focused on the top 3 interventions only, you'd get more than 80% of the real value of doing all 6. And if you focused on 3 instead of 6, it's much more likely that the patient would actually do them; you'd likely save a whole lot of money for both the patient and the payer; and you'd avoid the risk of medication side effects if the intervention involved a medicine. From both the individual patient-doc perspective, and the larger public health perspective, it's a smart move.
The drug industry, on the other hand, is much more likely to want docs to think that if the guidelines list 6 interventions, well doggone it, you had better do all 6. Recall that in our "diminishing turns" list for our patient with diabetes, the most effective intervention was the daily lo-dose aspirin, and the least was cholesterol lowering. To do the daily aspirin involves a cheap generic drug, that generates no profits to speak of for Pfizer or Merck or any of the big boys. (You don't see consumer ads on TV saying, "Ask your doctor if a daily generic aspirin tablet is right for you.") To get the man's cholsterol from 140 to 100 will probably take one if not two expensive statin drugs, many of which are still brand-name. Do you think the statin makers want us to think that cholesterol-lowering might be an expendible risk reduction in a smart patient-oriented strategy? Their marketing message to the doc is almost for sure to be that lowering cholesterol is every bit as important, if not more so, than any other risk reduction move.
A final tangent. Mold et al. give us pause not only about drug marketing; they also call into question the wisdom of most pay-for-performance strategies. In the abstract, the idea for paying the doc more for providing better quality care seems irrefutable. But if by "quality" we mean "meeting all the guideline targets" then we see how P4P can undermine smart practice. As the law of diminishing returns shows, you often do the best for your patient by getting close to, but not quite meeting the target--and adding more and more meds to be sure that you get to the target (whether cholesterol, or blood pressure, or blood sugar) might add very little extra benefit while greatly increasing the chances of adverse reactions. But the doc is heavily incentivized to try to meet the payment goal and not to think about what really most helps the patient.
Tip of the hat to Primary Care Medical Abstracts for including the Mold paper in their reviews.
Friday, January 14, 2011
The Inverse Benefit Law: Making Sense of How Marketing Turns (Maybe) Good Drugs into Bad
My esteemed colleague Donald Light, author/editor of the important volume, The Risks of Prescription Drugs (http://brodyhooked.blogspot.com/2010/08/how-many-new-drugs-are-lemons-ask.html) --to which I contributed a chapter--and I have just published a paper in American Journal of Public Health (subscription required). I immodestly believe that the concept in the paper is important enough to be worth laying out here in some detail.

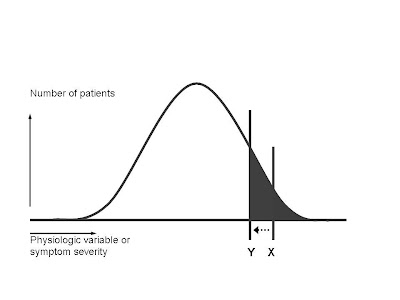
As you go to the right on the curve, the symptoms become more severe, or else the physiological variable that's supposed to be treated (such as blood sugar, cholesterol level, or blood pressure) becomes farther and farther above the mean for the population. As a general rule, the patients who can most benefit from a drug are clustered at the right-hand portion of the bell curve--those with the most severe symptoms, or the highest blood pressure, etc. By contrast, all patients anywhere under the curve are equally at risk for any adverse reactions the drug may cause.
 The line X demarcates an area under the curve (gray area) that represents the ideal benefit-harm ratio. The patients with the greatest need for the drug, who also have the greatest likelihood of benefiting from the drug, are supposed to receive it. (In statistical terms, this translates into a low number-needed-to-treat (NNT). NNT is like golf, low scores are better. It means that in order to get one patient to receive benefit, you have to treat a relatively small number of patients. The number of side effects is kept low, because a relatively small portion of the total population receives the drug. To the right of X, the benefit-harm ratio is quite favorable for the individual. They have a very good chance of receiving benefit, and that balances the real but small chance they might suffer an adverse reaction.
The line X demarcates an area under the curve (gray area) that represents the ideal benefit-harm ratio. The patients with the greatest need for the drug, who also have the greatest likelihood of benefiting from the drug, are supposed to receive it. (In statistical terms, this translates into a low number-needed-to-treat (NNT). NNT is like golf, low scores are better. It means that in order to get one patient to receive benefit, you have to treat a relatively small number of patients. The number of side effects is kept low, because a relatively small portion of the total population receives the drug. To the right of X, the benefit-harm ratio is quite favorable for the individual. They have a very good chance of receiving benefit, and that balances the real but small chance they might suffer an adverse reaction.
 The shape of the bell curve shows us why the "left shift" is a marketer's dream. You have moved the threshold line perhaps very little. (Let's say for example you decide that instead of trying to reach a target blood pressure of 140, you decide to try to reduce the blood pressure below 130.) But, if you do a little measuring, you'll see that in this hypothetical example (not related to blood pressure), you have increased the gray area under the curve by a factor of at least 4.
The shape of the bell curve shows us why the "left shift" is a marketer's dream. You have moved the threshold line perhaps very little. (Let's say for example you decide that instead of trying to reach a target blood pressure of 140, you decide to try to reduce the blood pressure below 130.) But, if you do a little measuring, you'll see that in this hypothetical example (not related to blood pressure), you have increased the gray area under the curve by a factor of at least 4.
This blog has addressed the many drug flops from recent years, where a drug introduced with much fanfare later turns out to be either much less effective, or much more dangerous, than first thought. The poster child is probably Vioxx, where the drug was actually removed from the market, but the list is long. The question we set out to address--is this random bad luck? Or is something going on systemically that accounts for this phenomenon?
Our answer is the Inverse Benefit Law: the ratio of benefits to harms among patients taking new drugs tends to vary inversely with how extensively the drugs are marketed. (Kudos to Don Light for thinking of this name for the law, which is a hat tip to the Inverse Care Law of Julian Tudor Hart.)
The Inverse Benefit Law seems counterintuitive for several reasons:
- The benefit-harm ratio of a drug should be dictated by the drug's properties, not how it is marketed.
- The drug industry's reputation is at stake: why wopuld they market drugs which are ineffective or unsafe?
- The FDA is supposed to protect us from ineffective or unsafe drugs.
So how could the Inverse Benefit Law possibly be correct? Let's walk through the reasoning.
First, let's assume that the target condition for a drug exists within the population and is distributed according to the infamous bell-shaped curve.

As you go to the right on the curve, the symptoms become more severe, or else the physiological variable that's supposed to be treated (such as blood sugar, cholesterol level, or blood pressure) becomes farther and farther above the mean for the population. As a general rule, the patients who can most benefit from a drug are clustered at the right-hand portion of the bell curve--those with the most severe symptoms, or the highest blood pressure, etc. By contrast, all patients anywhere under the curve are equally at risk for any adverse reactions the drug may cause.
Those assumptions tell us, as a general rule, where the scientifically evidence-based threshold will lie in recommending that physicians prescribe the drug:
 The line X demarcates an area under the curve (gray area) that represents the ideal benefit-harm ratio. The patients with the greatest need for the drug, who also have the greatest likelihood of benefiting from the drug, are supposed to receive it. (In statistical terms, this translates into a low number-needed-to-treat (NNT). NNT is like golf, low scores are better. It means that in order to get one patient to receive benefit, you have to treat a relatively small number of patients. The number of side effects is kept low, because a relatively small portion of the total population receives the drug. To the right of X, the benefit-harm ratio is quite favorable for the individual. They have a very good chance of receiving benefit, and that balances the real but small chance they might suffer an adverse reaction.
The line X demarcates an area under the curve (gray area) that represents the ideal benefit-harm ratio. The patients with the greatest need for the drug, who also have the greatest likelihood of benefiting from the drug, are supposed to receive it. (In statistical terms, this translates into a low number-needed-to-treat (NNT). NNT is like golf, low scores are better. It means that in order to get one patient to receive benefit, you have to treat a relatively small number of patients. The number of side effects is kept low, because a relatively small portion of the total population receives the drug. To the right of X, the benefit-harm ratio is quite favorable for the individual. They have a very good chance of receiving benefit, and that balances the real but small chance they might suffer an adverse reaction.The marketing folks at the drug company, however, have fits with drawing the line at X. It means that relatively few people in the entire population are "targets" for drug sales. Total revenues are very unlikely to reach the blockbuster status that shareholders demand.
So if you're a savvy drug marketer, what do you do? The best solution, from a revenue standpoint, is to try to do what we docs might call a "left shift"--move the threshold for prescribing the drug from X to Y:
Let's zoom in and take a closer look at the newly expanded gray area as a result of the "left shift":
 The shape of the bell curve shows us why the "left shift" is a marketer's dream. You have moved the threshold line perhaps very little. (Let's say for example you decide that instead of trying to reach a target blood pressure of 140, you decide to try to reduce the blood pressure below 130.) But, if you do a little measuring, you'll see that in this hypothetical example (not related to blood pressure), you have increased the gray area under the curve by a factor of at least 4.
The shape of the bell curve shows us why the "left shift" is a marketer's dream. You have moved the threshold line perhaps very little. (Let's say for example you decide that instead of trying to reach a target blood pressure of 140, you decide to try to reduce the blood pressure below 130.) But, if you do a little measuring, you'll see that in this hypothetical example (not related to blood pressure), you have increased the gray area under the curve by a factor of at least 4. For marketing purposes this is great--you have 4 times as many folks out there to whom you can try to sell your drug (either by the reps marketing to their docs, or by direct-to-consumer ads).
For purposes of improving the health of the population, it's a disaster. NNT goes way up-- you have to treat many more patients for a single one to benefit. The number of patients sufferuing adverse reactions goes up by the same factor of 4. The ratio of benefit to harm turns very negative.
So--how can the marketers make this left shift happen? We go into several mechanisms in the article, and give specific examples of which drugs have recently been marketed by each technique. None of these techniques will be strangers to regular readers of this blog. Briefly, we discuss: reducing the thresholds for diagnosing the disease (the magic cutoff for diabetes used to be blood sugar of 140, then 126, and finally 110, with the guidelines suggesting these changes often being written by docs with financial ties to drugmakers); using surrogate endpoints such as blood pressure, blood sugar, etc. to sell drugs that don't change the outcomes really important to patients like preventing stroke, heart attacks, and deaths; exaggerating benefits; minimizing adverse effects; creating new diseases like "social phobia" or making previous non-disease states into diseases (like osteopenia); and promoting off-label prescribing.
We then run the drill about what needs to be done to halt each type of marketing ploy, and to remind docs that they are supposed to treat patients first and not become the tools of the industry's marketing mechanism.
I believe our single most important line is: "Our claim, therefore, is not that the pharmaceutical industry sets out to market bad drugs. Rather, the scientific arm of the industry works hard to discover new drugs that are both effective and safe. The marketing arm then turns those good drugs into bad drugs, in effect, by extending their use beyond the proper evidence base."
I believe our single most important line is: "Our claim, therefore, is not that the pharmaceutical industry sets out to market bad drugs. Rather, the scientific arm of the industry works hard to discover new drugs that are both effective and safe. The marketing arm then turns those good drugs into bad drugs, in effect, by extending their use beyond the proper evidence base."
Obviously, Don and I thought that this heuristic device (we never intended the Inverse Benefit "Law" to function as any sort of precise mathematical calculation) is generally accurate and illustrative. I ask readers of the blog to point out flaws and weaknesses in this concept. (In my next few posts I'll review another recent paper that seems to offer indirect support to the Inverse Benefit Law.)
Brody H, Light DW. The inverse benefit law: how drug marketing undermines patient safety and public health. American Journal of Public Health doi:10.2105/AJPH.2010.199844, epub Jan. 13, 2011.
Hart JT. The inverse care law. Lancet 1(7696):405-12, 1971.
Friday, January 7, 2011
Paying Attention to the Man Behind the Curtain: KOL Handlers for Hire
Yes, I know, maybe this blog and Health Care Renewal should simply merge and be done with it. But I was quite impressed with Roy Poses' detective work:
http://hcrenewal.blogspot.com/2011/01/key-opinion-leader-services-companies.html
Dr. Poses was checking out some facts related to another issue when he came across mention of a type of firm neither he nor I had previous heard of. He here posts his findings about three commercial outfits that cater to drug companies and similar firms, and offer their services to find and manage key opinion leaders (KOLs) in medicine.
Dr. Poses's main point is that these KOLs continue to imagine that the drug companies want their expertise and opinions, when in fact the companies regard them as marketing devices pure and simple. (Or, as in his polite phrase, "idiots.") The marketing stuff that the KOL handlers use on the drug companies to peddle their services try to sugar-coat it as much as they can, but it takes little sophistication to read between the lines and see that they are appealing purely to sales potential.
The implicit message in these marketing messages seem to be that you can view KOLs as either sheep or goats. Sheep are like, well, sheep. You herd them and then you shear them. The handling firms promise that they'll get you a whole lot of sheep, and herd them in the direction you want them to go. Goats, on the other hand, are pesky critters with much more of a mind of their own. They can be troublesome and get into things they're not supposed to. The handling firms promise that if you are dealing with the goat species of KOL, they'll help you to manage them so that they stay out of trouble.
The comment that I would add to this comes from HOOKED, where I belatedly learned that if you check out the website of a large medical meeting (in my case, the annual meeting of the American Academy of Family Physicians, my own specialty society), you'll see that it is really two parallel websites. There is one for the docs and another for the advertisers and others who want to rent booths and sell their wares. The doc website is what you'd expect, all about the great things you'll learn, plus of course the fun you can have visiting the city where the meeting is held, especially if youbring your family. The advertiser website is another beast entirely. It basically says that we have this captive audience of docs, all of whom we can lead around by the nose at will, and if you rent a booth in our exhibit hall, we'll lead all those docs direct to your booth, and then your sales will skyrocket. Plus when those docs go home they'll tell all their envious colleagues, who did not get to go to our meeting, what they saw at your booth, and then your sales will skyrocket even more.
It seems simply to be assumed that the docs will never read the advertisers' parallel website.
http://hcrenewal.blogspot.com/2011/01/key-opinion-leader-services-companies.html
Dr. Poses was checking out some facts related to another issue when he came across mention of a type of firm neither he nor I had previous heard of. He here posts his findings about three commercial outfits that cater to drug companies and similar firms, and offer their services to find and manage key opinion leaders (KOLs) in medicine.
Dr. Poses's main point is that these KOLs continue to imagine that the drug companies want their expertise and opinions, when in fact the companies regard them as marketing devices pure and simple. (Or, as in his polite phrase, "idiots.") The marketing stuff that the KOL handlers use on the drug companies to peddle their services try to sugar-coat it as much as they can, but it takes little sophistication to read between the lines and see that they are appealing purely to sales potential.
The implicit message in these marketing messages seem to be that you can view KOLs as either sheep or goats. Sheep are like, well, sheep. You herd them and then you shear them. The handling firms promise that they'll get you a whole lot of sheep, and herd them in the direction you want them to go. Goats, on the other hand, are pesky critters with much more of a mind of their own. They can be troublesome and get into things they're not supposed to. The handling firms promise that if you are dealing with the goat species of KOL, they'll help you to manage them so that they stay out of trouble.
The comment that I would add to this comes from HOOKED, where I belatedly learned that if you check out the website of a large medical meeting (in my case, the annual meeting of the American Academy of Family Physicians, my own specialty society), you'll see that it is really two parallel websites. There is one for the docs and another for the advertisers and others who want to rent booths and sell their wares. The doc website is what you'd expect, all about the great things you'll learn, plus of course the fun you can have visiting the city where the meeting is held, especially if youbring your family. The advertiser website is another beast entirely. It basically says that we have this captive audience of docs, all of whom we can lead around by the nose at will, and if you rent a booth in our exhibit hall, we'll lead all those docs direct to your booth, and then your sales will skyrocket. Plus when those docs go home they'll tell all their envious colleagues, who did not get to go to our meeting, what they saw at your booth, and then your sales will skyrocket even more.
It seems simply to be assumed that the docs will never read the advertisers' parallel website.
Thursday, January 6, 2011
Dr. Steve Nissen on Lack of New Drugs
Thanks to a faithful reader for calling my attention to this nice 6-minute video interview with Dr. Steve Nissen, cardiologist at Cleveland Clinic:
http://www.washingtonpost.com/wp-dyn/content/video/2011/01/06/VI2011010603537.html
The reporter for Bloomberg asks Dr. Nissen--isn't it bad for medicine that drug firms are in jeopardy of losing profits in coming years due to a shrinking dug pipeline and fewer new drugs approved in 2010 than in recent memory? Isn't this a sign that the FDA is being too obstructionist in approving new drugs?
Dr. Nissen, bless him, is having none of this. Better to have fewer, better, and safer drugs, he says. He offers as two reasons for the dwindling drug pipeline--first, the low hanging fruit theory that I favor in HOOKED--that with so many useful drugs having already been discovered, it is harder and harder to find new ones that are equally good and also safe. (He could have said it a bit more starkly--what the heck ever persuaded anyone that real scientific breakthroughs could be timed to occur precisely in accord with the drug companies' budget and patent cycles?) Second, he points out that increasingly the industry does not invest in real innovation, but instead "me too" drugs that can most quickly be marketed. If you don't invest in innovation, don't be surprised if you don't have any, says Dr. Nissen.
Then he goes on as a bonus to say a few choice things about critics of health reform. But that's not what this blog is about.
http://www.washingtonpost.com/wp-dyn/content/video/2011/01/06/VI2011010603537.html
The reporter for Bloomberg asks Dr. Nissen--isn't it bad for medicine that drug firms are in jeopardy of losing profits in coming years due to a shrinking dug pipeline and fewer new drugs approved in 2010 than in recent memory? Isn't this a sign that the FDA is being too obstructionist in approving new drugs?
Dr. Nissen, bless him, is having none of this. Better to have fewer, better, and safer drugs, he says. He offers as two reasons for the dwindling drug pipeline--first, the low hanging fruit theory that I favor in HOOKED--that with so many useful drugs having already been discovered, it is harder and harder to find new ones that are equally good and also safe. (He could have said it a bit more starkly--what the heck ever persuaded anyone that real scientific breakthroughs could be timed to occur precisely in accord with the drug companies' budget and patent cycles?) Second, he points out that increasingly the industry does not invest in real innovation, but instead "me too" drugs that can most quickly be marketed. If you don't invest in innovation, don't be surprised if you don't have any, says Dr. Nissen.
Then he goes on as a bonus to say a few choice things about critics of health reform. But that's not what this blog is about.
'Tis a French Custom--Libel Suits?
Q: How does a smart drug company discourage a non-commercial evidence-based information source from saying true but unfavorable things about one of their products?
A: Sue them for libel.
At least if you're in France.
http://www.ahrp.org/cms/content/view/748/9/ explains how Astellas Pharma, a Japanese-based multinational, is suing the noted French periodical Prescrire for libel over the publication's discouraging review of their drug, Protopic, an anti-eczema ointment which according to an FDA warning carries a cancer risk due to its immune suppression.
Dubois and Braillon, writing in the UK's HealthWatch, explain how France has become a sort of haven for these libel suits, with French institutions often being slow to defend a scientist who's attacked in this way: http://href.fr/healthwatch_oct10.pdf
In HOOKED I described suits of this sort as a variation on what's called SLAPP (strategic lawsuit against public participation). The basic idea behind SLAPP is that the plaintiff has very little if any chance of winning the suit; but the plan is both to impoverish the relatively impecunious defendent by running up a big legal bill to defend themselves against a deep-pockets company that has stables of lawyers on retainer, and indirectly to intimidate others from speaking out against the company for fear of similar treatment.
And here I thought that the US was the world capital for excessive litigation...
A: Sue them for libel.
At least if you're in France.
http://www.ahrp.org/cms/content/view/748/9/ explains how Astellas Pharma, a Japanese-based multinational, is suing the noted French periodical Prescrire for libel over the publication's discouraging review of their drug, Protopic, an anti-eczema ointment which according to an FDA warning carries a cancer risk due to its immune suppression.
Dubois and Braillon, writing in the UK's HealthWatch, explain how France has become a sort of haven for these libel suits, with French institutions often being slow to defend a scientist who's attacked in this way: http://href.fr/healthwatch_oct10.pdf
In HOOKED I described suits of this sort as a variation on what's called SLAPP (strategic lawsuit against public participation). The basic idea behind SLAPP is that the plaintiff has very little if any chance of winning the suit; but the plan is both to impoverish the relatively impecunious defendent by running up a big legal bill to defend themselves against a deep-pockets company that has stables of lawyers on retainer, and indirectly to intimidate others from speaking out against the company for fear of similar treatment.
And here I thought that the US was the world capital for excessive litigation...
Wednesday, January 5, 2011
What Do Pharma Execs Think?
Sorry to bore you with yet another tip of the hat to my esteemed colleague Roy Poses over at the Health Care Renewal blog, but I owe it to him that I am aware of this web page:
http://blog.pharmexec.com/2011/01/05/new-year-night-stalkers-what-will-keep-the-c-suiters-awake-in-2011/
So-- how does PharmExec.com see the world today? One thing I find intriguing is that we pharmascold types think of the drug industry as this huge, powerful juggernaut--as pointed out by Marcia Angell, if we go back to the olden days of 2002, even though major drug firms made up only 2% of the Fortune 500 companies, their total profits were more than half of the total profits of all 500 firms. Yet whenever we get to look through the keyhole at what Pharma execs think is going on, they see themselves as being beleaguered by all sorts of forces out to get them, and the survival of their companies from one year to the next has to be regarded as near-miraculous.
Here's how the post starts: "The consensus is that 2011 will be a bad year for Big Pharma. It must confront a breaking wave of patent expirations, while fiscal retrenchment has created an innovation cycle in reverse as payers find new ways to curb the drugs bill. Risk-averse regulators are transforming old tools like the FDA “complete response letter” into a registration parking lot, with no exit ramp to connect companies to a distracted — and increasingly impatient — community of clinicians and consumers."
Let's make a short list.
Yet it would be terribly short-sighted to dismiss this inside view as mere victimhood wailing. Here and there the post gives a little bit of evidence that even the suits now realize that all is not right with how they do business.
http://blog.pharmexec.com/2011/01/05/new-year-night-stalkers-what-will-keep-the-c-suiters-awake-in-2011/
So-- how does PharmExec.com see the world today? One thing I find intriguing is that we pharmascold types think of the drug industry as this huge, powerful juggernaut--as pointed out by Marcia Angell, if we go back to the olden days of 2002, even though major drug firms made up only 2% of the Fortune 500 companies, their total profits were more than half of the total profits of all 500 firms. Yet whenever we get to look through the keyhole at what Pharma execs think is going on, they see themselves as being beleaguered by all sorts of forces out to get them, and the survival of their companies from one year to the next has to be regarded as near-miraculous.
Here's how the post starts: "The consensus is that 2011 will be a bad year for Big Pharma. It must confront a breaking wave of patent expirations, while fiscal retrenchment has created an innovation cycle in reverse as payers find new ways to curb the drugs bill. Risk-averse regulators are transforming old tools like the FDA “complete response letter” into a registration parking lot, with no exit ramp to connect companies to a distracted — and increasingly impatient — community of clinicians and consumers."
Let's make a short list.
- We say: Even in European countries with price controls, that pay only about two-thirds of what the US pays for drugs, companies make plenty of profit to support R&D; and there's no serious drug cost containment on the horizon in the US. They say: Today, no country feels that it ought to have to pay for drug R&D, so the industry will soon find its research funds drying up.
- We say: It's a great step forward when resource-poor countries can negotiate a deal with a multinational drug firm for manufacturing of the drug to occur in a local factory, assuring both a more affordable drug for the local population plus jobs for the local economy. They say: That emerging-nation firm that they've been forced to work with is a potential future competitor, and they've been forced to grant that competitor substantial inside know-how in drug manufacturing--what amounts to the theft of intellectual property.
Yet it would be terribly short-sighted to dismiss this inside view as mere victimhood wailing. Here and there the post gives a little bit of evidence that even the suits now realize that all is not right with how they do business.
- On the incredible amount ($10-14B, depending on who you ask) that Pharma has paid out recently in criminal settlements: "Despite this, pharma has failed to address the problem from an industry-wide reputational, as opposed to a legal, standpoint. ”Tops in Fraud” is a ruinous moniker for a business so dependent on basic issues of trust like integrity, quality and safety – when will the industry, as part of a collective action, replace the gamey politicking with good policing?"
- On the relentless outsourcing of research to cut costs and avoid ethical scrutiny: "The traditional business model of Big Pharma — with its heavy investment in in-house innovation – is being reconsidered through new approaches that emphasize the outsourcing of R&D: from research to “search,” with the latter linked to external licensing and partnering. Taken to its logical extent, the new model could transform companies from innovators to distribution platforms that rely on marketing heft, size and scale to compete rather than science. Coupled with the ruthless drive for efficiencies that have led to large-scale layoffs of once cosseted professional staff, the trend raises an important reputational question: if the industry is no longer viewed as a wellspring of science and innovation, what strategy is in play to respond to a world that perceives industry as the WalMart of pills?"
- On take-no-prisoners marketing battles: "Stiff competition within therapeutic classes has created unheard of rivalries among companies that once were happy to share the same watering hole. Some marketers are investing millions in brand-bashing “anti-launch” strategies to limit the uptake of newer alternative products. The question is whether everyone loses when this logic is applied against the larger patient-first perspective that regulators and the public expect from the industry."
New Proposals for CME Reforms
Health law scholar Marc A. Rodwin of Suffolk U. Law School provides a very useful historical overview of drug industry involvement in continuing medical education and then offers some novel ideas for reform (subscription required).
In HOOKED I harp on the lack of historical perspective that infects most recent work on the pharmaceutical industry, and often portrays all the serious problems at the medicine-Pharma interface as if they first cropped up yesterday. Hence any thoughtful historical reflection is welcome, and Rodwin provides a comprehensive review of how drug advertising and CME evolved hand in glove through the 20th century.
Rodwin conducted interviews with CME managers, allowing him to make statements that put the lie to the comforting mantra that industry-sponsored CME is really education and not marketing. Perhaps the single most telling quote of this type is: "Moreover, CME providers traditionally sought funds from one firm per program because, as a manager of a CME provider explained to me, drug firms believed a provider supported by two or more firms with competing drugs would have a conflict of interest." Notice the obvious: if a conference were truly about education and not about pushing product, then having multiple industry sponsors would seem completely benign and indeed desirable. Also, as a secondary issue, notice that the naysayers who deny that "conflict of interest" exists or is a truly meaningful term of professional ethics, are apparently not joined by those within Pharma itself!
Rodwin joins those who criticized the 2004 ACCME guidelines as too wimpy--he notes that the rules say that a CME provider "cannot be required" by a sponsor to accept advice or direction regarding speakers or program content, meaning, "That language does not prohibit commercial supporters from offering advice, CME providers from soliciting suggestions from them, or CME providers voluntarily following suggestions of commercial supporters. My interviews with CME providers indicated that these were common practices."
So--what to do? Rodwin starts with a novel proposal. How about CME folks actually having to decide upon a curriculum? A medical school or a residency program that did not teach in accord with a standard curriculum decided upon by consensus within the relevant community of experts would speedily lose accreditation. Why, then, allow CME programs to be driven by the whims of who wants to speak on which topic, or which company wants to pay the freight? Merely having an agreed-upon curriculum based on science and real practice issues would go a long way toward correcting the tilt toward talking only about drugs, devices, and stuff that makes money for industry.
Past that, Rodwin would ban all direct and most indirect industry funding of CME, and ask Congress to impose a CME tax on all those who currently make profits off medical care (docs included). (Rodwin treats the idea that merely disclosing industry sponsorship of a CME program solves anything at all as hardly worth discussing.) If the anti-tax people start to howl about this, Rodwin reminds us that commercial firms that now pay for CME pass the resulting costs through to the end-purchaser anyway, so we all end up paying. If we did it via the tax, and allocated the tax proceeds to a federal agency who distributed them to non-profit CME entities, we'd have a greater likelihood that the same money would actually go toward improving our health.
Rodwin MA. Drug advertising, continuing medical education, and physician prescribing: a historical review and reform proposal. Journal of Law, Medicine and Ethics 38:807-815, Winter 2010.
In HOOKED I harp on the lack of historical perspective that infects most recent work on the pharmaceutical industry, and often portrays all the serious problems at the medicine-Pharma interface as if they first cropped up yesterday. Hence any thoughtful historical reflection is welcome, and Rodwin provides a comprehensive review of how drug advertising and CME evolved hand in glove through the 20th century.
Rodwin conducted interviews with CME managers, allowing him to make statements that put the lie to the comforting mantra that industry-sponsored CME is really education and not marketing. Perhaps the single most telling quote of this type is: "Moreover, CME providers traditionally sought funds from one firm per program because, as a manager of a CME provider explained to me, drug firms believed a provider supported by two or more firms with competing drugs would have a conflict of interest." Notice the obvious: if a conference were truly about education and not about pushing product, then having multiple industry sponsors would seem completely benign and indeed desirable. Also, as a secondary issue, notice that the naysayers who deny that "conflict of interest" exists or is a truly meaningful term of professional ethics, are apparently not joined by those within Pharma itself!
Rodwin joins those who criticized the 2004 ACCME guidelines as too wimpy--he notes that the rules say that a CME provider "cannot be required" by a sponsor to accept advice or direction regarding speakers or program content, meaning, "That language does not prohibit commercial supporters from offering advice, CME providers from soliciting suggestions from them, or CME providers voluntarily following suggestions of commercial supporters. My interviews with CME providers indicated that these were common practices."
So--what to do? Rodwin starts with a novel proposal. How about CME folks actually having to decide upon a curriculum? A medical school or a residency program that did not teach in accord with a standard curriculum decided upon by consensus within the relevant community of experts would speedily lose accreditation. Why, then, allow CME programs to be driven by the whims of who wants to speak on which topic, or which company wants to pay the freight? Merely having an agreed-upon curriculum based on science and real practice issues would go a long way toward correcting the tilt toward talking only about drugs, devices, and stuff that makes money for industry.
Past that, Rodwin would ban all direct and most indirect industry funding of CME, and ask Congress to impose a CME tax on all those who currently make profits off medical care (docs included). (Rodwin treats the idea that merely disclosing industry sponsorship of a CME program solves anything at all as hardly worth discussing.) If the anti-tax people start to howl about this, Rodwin reminds us that commercial firms that now pay for CME pass the resulting costs through to the end-purchaser anyway, so we all end up paying. If we did it via the tax, and allocated the tax proceeds to a federal agency who distributed them to non-profit CME entities, we'd have a greater likelihood that the same money would actually go toward improving our health.
Rodwin MA. Drug advertising, continuing medical education, and physician prescribing: a historical review and reform proposal. Journal of Law, Medicine and Ethics 38:807-815, Winter 2010.
Monday, January 3, 2011
Nurse Practitioners: The Next Frontier?
Elissa Ladd and colleagues from Harvard have published a study:
http://www.ajmc.com/issue/managed-care/2010/2010-12-vol16-n12/AJMC_10decLadd_WebX_e358to62
--on attitudes among a national sample of nurse practitioners regarding interactions with the drug industry. We have seen reason to believe that in recent years, physicians have become at least slightly more skeptical regarding industry marketing overtures (http://brodyhooked.blogspot.com/2010/11/are-physicians-taking-fewer-bribesgifts.html). We have also seen some evidence that the industry has increasingly targeted nurse practitioners, who have prescribing privileges (http://brodyhooked.blogspot.com/2008/02/nurses-soft-targets-for-pharma.html and http://brodyhooked.blogspot.com/2007/10/if-you-dont-feed-them-they-wont-come.html). Ladd's group did the first extensive national survey, which was marred by a very low response rate of 9%, though not that atypical of online surveys. If we can assume their sample was somewhat representative, a number of worrisome conclusions emerge:
Finally, it is worrisome, as Ladd and colleagues point out, that while the new sunshine provisions of the health reform law (PPACA) require as of 2013 that all payments to physicians be reported, payments to NPs are not included under its provisions.
http://www.ajmc.com/issue/managed-care/2010/2010-12-vol16-n12/AJMC_10decLadd_WebX_e358to62
--on attitudes among a national sample of nurse practitioners regarding interactions with the drug industry. We have seen reason to believe that in recent years, physicians have become at least slightly more skeptical regarding industry marketing overtures (http://brodyhooked.blogspot.com/2010/11/are-physicians-taking-fewer-bribesgifts.html). We have also seen some evidence that the industry has increasingly targeted nurse practitioners, who have prescribing privileges (http://brodyhooked.blogspot.com/2008/02/nurses-soft-targets-for-pharma.html and http://brodyhooked.blogspot.com/2007/10/if-you-dont-feed-them-they-wont-come.html). Ladd's group did the first extensive national survey, which was marred by a very low response rate of 9%, though not that atypical of online surveys. If we can assume their sample was somewhat representative, a number of worrisome conclusions emerge:
- 96% have regular contact with sales reps
- 83% think information provided by reps is reliable
- 93% believe themselves uninfluenced by reps' free gifts
- 78% think free meals provided by industry are a good/excellent way to learn about new drugs
- 83% think industry-sponsored continuing education is a good/excellent way to provide reliable education at affordable cost
- 90% think it ethically acceptable to attend industry sponsored meal events
- 75% think it ethically acceptable for speakers to be paid by drug companies
- 61% think taking small gifts and meals is ethically acceptable
Finally, it is worrisome, as Ladd and colleagues point out, that while the new sunshine provisions of the health reform law (PPACA) require as of 2013 that all payments to physicians be reported, payments to NPs are not included under its provisions.
Saturday, January 1, 2011
The Anatomy of Spin
An article (subscription required) that was published some months ago provides some useful information about spin in medical research publications, raises some important qustions, and poses a mystery of its own.
A group out of Oxford and Paris set out to describe exactly how spin appears in medical research publications. They searched databases for articles published between December 2006 and March 2007 reporting the results of randomized controlled trials. They were on the lookout for articles that reported trials in which the difference in the primary outcomes was statistically insignificant--figuring that this was the sort of paper where the temptation to introduce spin was greatest, and hence these papers would be most useful to decide what forms the spin took. They started with 1735 potentially applicable titles but ended up with only 72 papers that met all their criteria. They then proceeded to develop an assessment of the presence, degree, and categories of spin, which of necessity was a subjective enterprise. They also noted that in only 44 of the 72 papers were the primary research outomes clearly identified.
They found that among the 72 papers, 33% were funded wholly or in part by for-profit entities, and in another 37.5% the source of funding was not reported.
They found that spin was common--40% of the papers had spin in at least two separate sections of the main text, and 58% had spin in the Conclusion section of the abstract. The forms that the spin took included focusing on other results (such as within-group comparisons) that were statistically significant while downplaying the lack of significance in the primary outcomes; interpreting the lack of statistical significance as showing equivalence ("at least our treatment was shown to be no worse than...") which is a huge methodological no-no; and simply ignoring the lack of significance and playing up the supposed benefits and/or safety of the experimental treatment anyway.
So now we come to the mystery: the authors never reported how the level of spin correlated with for-profit funding. At one point they state, "Our results are consistent with those of other related studies showing a positive relation between financial ties and favorable conclusions stated in trial reports." But they give no numbers anywhere. My only hunch as to why they report no data on this apparently key variable is that with only 72 studies in their final sample, the numbers would have been too small to be of much reliability, and they did not want to be charged with committing themselves the very spin they were criticizing. But one would have expected an explanation of some sort.
OK, so what else can we glean from this study? Mainly one thing. Where the heck are the journal editors and reviewers? How can a study be published in a supposedly respectable journal, within the past few years, that fails to specify any source of funding for a major clinical trial, or that fails to make clear what are the primary outcomes? Plus, these authors were able to detect spin in a substantial percentage of these papers, based solely on the contents of the paper--they sought no access to any other data. If these authors could, the journal editors and reviewers presumably could too--yet they let these blatant misstatements pass. In short, if industry sponsors of research are seeking to add spin to their publications to goose the marketing of their products, it seems that today's journal editing apparatus is putty in their hands.
This might seem to be unfair criticism because the authors focused on only one specific type of research report--an RCT where the result is insignificant, but that is published anyway. (The problem of journals refusing to publish papers that produce non-statistically-significant findings, thereby skewing the publication record, is another issue beyond our scope here.) It may be that a lot of spin appears in such reports, but very little spin appears in reports of RCTs where the results reach statistical significance--no suprise if so, as there's then much less need to add spin. OK, fair enough. But if these authors focused especially on that sort of trial report because they expected that setting to create a strong temptation to add spin, then journal editors and reviewers should have been equally forewarned and should have been especially vigilant. So the fact that so much spin and nonreporting still made it through the review process is very worrisome.
Boutron I, Dutton S, Ravaud P, Altman DG. Reporting and interpretation of randomized controlled trials with statistically nonsignificant results for primary outcomes. JAMA 303:2058-64, May 26, 2010.
A group out of Oxford and Paris set out to describe exactly how spin appears in medical research publications. They searched databases for articles published between December 2006 and March 2007 reporting the results of randomized controlled trials. They were on the lookout for articles that reported trials in which the difference in the primary outcomes was statistically insignificant--figuring that this was the sort of paper where the temptation to introduce spin was greatest, and hence these papers would be most useful to decide what forms the spin took. They started with 1735 potentially applicable titles but ended up with only 72 papers that met all their criteria. They then proceeded to develop an assessment of the presence, degree, and categories of spin, which of necessity was a subjective enterprise. They also noted that in only 44 of the 72 papers were the primary research outomes clearly identified.
They found that among the 72 papers, 33% were funded wholly or in part by for-profit entities, and in another 37.5% the source of funding was not reported.
They found that spin was common--40% of the papers had spin in at least two separate sections of the main text, and 58% had spin in the Conclusion section of the abstract. The forms that the spin took included focusing on other results (such as within-group comparisons) that were statistically significant while downplaying the lack of significance in the primary outcomes; interpreting the lack of statistical significance as showing equivalence ("at least our treatment was shown to be no worse than...") which is a huge methodological no-no; and simply ignoring the lack of significance and playing up the supposed benefits and/or safety of the experimental treatment anyway.
So now we come to the mystery: the authors never reported how the level of spin correlated with for-profit funding. At one point they state, "Our results are consistent with those of other related studies showing a positive relation between financial ties and favorable conclusions stated in trial reports." But they give no numbers anywhere. My only hunch as to why they report no data on this apparently key variable is that with only 72 studies in their final sample, the numbers would have been too small to be of much reliability, and they did not want to be charged with committing themselves the very spin they were criticizing. But one would have expected an explanation of some sort.
OK, so what else can we glean from this study? Mainly one thing. Where the heck are the journal editors and reviewers? How can a study be published in a supposedly respectable journal, within the past few years, that fails to specify any source of funding for a major clinical trial, or that fails to make clear what are the primary outcomes? Plus, these authors were able to detect spin in a substantial percentage of these papers, based solely on the contents of the paper--they sought no access to any other data. If these authors could, the journal editors and reviewers presumably could too--yet they let these blatant misstatements pass. In short, if industry sponsors of research are seeking to add spin to their publications to goose the marketing of their products, it seems that today's journal editing apparatus is putty in their hands.
This might seem to be unfair criticism because the authors focused on only one specific type of research report--an RCT where the result is insignificant, but that is published anyway. (The problem of journals refusing to publish papers that produce non-statistically-significant findings, thereby skewing the publication record, is another issue beyond our scope here.) It may be that a lot of spin appears in such reports, but very little spin appears in reports of RCTs where the results reach statistical significance--no suprise if so, as there's then much less need to add spin. OK, fair enough. But if these authors focused especially on that sort of trial report because they expected that setting to create a strong temptation to add spin, then journal editors and reviewers should have been equally forewarned and should have been especially vigilant. So the fact that so much spin and nonreporting still made it through the review process is very worrisome.
Boutron I, Dutton S, Ravaud P, Altman DG. Reporting and interpretation of randomized controlled trials with statistically nonsignificant results for primary outcomes. JAMA 303:2058-64, May 26, 2010.
Subscribe to:
Comments (Atom)